|
| Neither a scientist nor a cartoonist, I humbly present to you my drawing of a water molecule, known as H20. It is also known as the "Mickey Mouse" molecule due to not to its moles (hahahaha!) , but to its big "ears" of hydrogen. |
I spent last week spinning around with the electrons in the hydrogen and oxygen atoms, trying to understand how clouds become visible, which is to say how water molecules scatter light. All day last Friday, I drew diagrams of oxygen and hydrogen electron shells in my notebook. I moved their electrons and bonded them together into water molecules. I drew oscillating arrows the colors of visible light (your grade-school pal, ROY G BIV--red, orange, yellow, green, blue, indigo, violet).
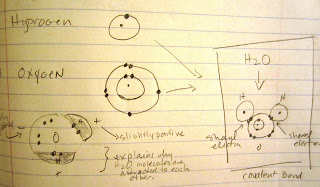 |
| I am pretty sure I did this same drawing in my 6th-grade science class. Luckily, nothing has changed in 40 years. |
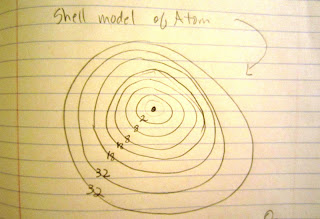 |
| I would throwing darts at this had I not labeled my diagram. This is the planetary model of the atom. This is just one idea, one graspable and graphical idea, of the inner workings of an atom. It was developed by Neils Bohr (1885-1962). The numbers represent the maximum number of electrons in each ring. |
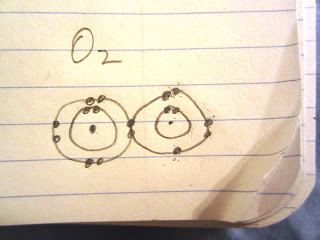 |
| Shell shocked: I got so dizzy bonding these two oxygen atoms that I lost track of their electrons. I think I have created something atomically impossible, illegal, ionic, and possibly quite dangerous. |
I had read that air molecules "selectively scatter" certain the shorter wavelengths of visible light to give us our blue skies and that a cloud droplet scatters all the wavelengths equally to give us our white clouds. But for as many times as I have read the explanations and studied the diagrams in my meteorology and chemistry text books, I still couldn't quite piece together and hold on to the whole story. I realized I needed help.
As soon as my husband (aka Dr. Science) walked in from work Friday night, I pounced. I put my my hands, clenched into fists, on the top of my head where ears would be if I were a mouse.
"Okay," I said. "I am a water molecule. My head is oxygen, my fists are hydrogen. You are visible light. Explain to me how light waves scatter."
Bless his heart, he went with it. He pretended his hands were short waves (G BIV) and his arms the long waves (ROY) bouncing off or being absorbed by the water molecule. Alas, I have no photograph to post.
What I understood from our molecular pantomime was that within the spectrum of visible light, there are waves of different lengths and frequencies. The shorter wavelengths had higher frequency and more energy; the longer wavelengths had lower frequency and less energy. When these wavelengths entered the atmosphere and encountered nitrogen, oxygen, water, and other molecules, they responded to the energy levels of those molecules. Those energy levels were determined by the number of electrons in the atoms' orbits.
I have been thinking about how to tell this now-subatomic story. It will take me a while but will appear as Part Two of "Why Clouds are White." Meanwhile, here is a limited edition jpg of pure white light being scattered in a sky blue and white cloud.
 |
| Do you know how hard it is to draw white light on white paper? |
Okay, my psychedelic drawing doesn't look like sunlight, a blue sky, or a white cloud. It does, however, illustrate how deeply I do not understand how light is scattered. Oh, but I will.
 |
| Possible sky color (center) for Earth when crayons instead of electromagnetic radiation are involved. |